FDA suggests restructuring food safety efforts into new human foods program
Human Foods Advisory Committee will advise on food safety, nutrition, technology issues.
courtesy of: FDA
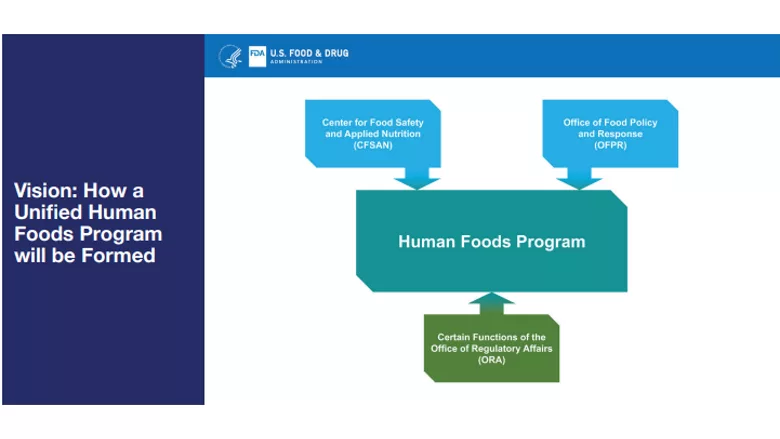
In a January 31 announcement, FDA proposed a vision to restructure the oversight of food safety and informed consumer choices into a new organization which will be known as the Human Foods Program. As part of this restructuring, the Center for Food Safety and Applied Nutrition (CFSAN), the Office of Food Policy and Response (OFPR), and portions of the Office of Regulatory Affairs (ORA) will be realigned as the agency continues working towards the promise of a New Era of Smarter Food Safety.
Included in this restructuring will be a Human Foods Advisory Committee, consisting of external partners that will advise on challenging and emerging issues in food safety, nutrition, and innovative technologies. It will be supported by improved analytical capabilities, risk algorithms, operational efficiencies, and streamlined communications.
Particularly notable to food safety is the establishment of an Office of Integrated Food Safety System Partnerships that will elevate, coordinate, and integrate food safety and response activities with state and local regulatory partners. Originally envisioned in the Food Safety Modernization Act (FSMA), this office will enhance collaboration with all stakeholders.
Additionally, a new Center for Excellence in Nutrition and proposed establishment of an Office of Critical Foods as directed by the 2023 Consolidated Appropriations Act will prioritize ongoing efforts that help consumers make more informed food choices and will work with industry to offer healthier and more nutritious food products.
The FDA’s lead office for all agency field activities, Office of Regulatory Affairs (ORA), operating system restructuring will enable better focus on critical activities related to its core mission of protecting public health through inspections, laboratory testing, imports, and investigations.
These changes are in response to an evaluation and findings of an external expert panel, in response to the infant formula supply chain issues, as well as recommendations by the agency’s own “boots on the ground” who work to ensure the safety of the food supply in the US.
The new Human Foods Program will be overseen by the newly created position of Deputy Commissioner for Human Foods, who will report to FDA Commissioner, Robert M. Califf, M.D. This person will unify and elevate the Human Food Program while removing redundancies and enabling the agency to oversee human food in a more effective and efficient way.
Additionally, the Center for Veterinary Medicine (CVM) will see a newly created Office of Animal Biotechnology Innovation which will enable robust cross collaboration between CVM and the Human Foods Program on agricultural biotechnology innovation.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






